Molecular orbital diagram of o29/10/2023  .png)
It is formed by removing 2 electrons from helium atom. Atoms are the smallest units of matter that can take part in a chemical reaction.Īn α (alpha) particle is a doubly charged helium ion (He 2+) containing two protons and two neutrons.Atoms of one element combine with atoms of another element in a simple ratio to form molecules of compounds.Atoms of an element combine in small numbers to form molecules.The atoms of an element are alike in all respects, but they differ from the atoms of other elements.Atoms can neither be created nor be destroyed.Matter consists of very small and indivisible particles called atoms.Main postulates of Dalton's atomic theory are: The existence of different types of matter is considered to be due to different types of atoms constituting them. He considered atoms as indivisible particles that are the fundamental building blocks of matter. John Dalton gave the first scientific theory about the structure of matter. (b) Democritus called the paramanu an atom, which is derived from the Greek word atomos, meaning indivisible. Each one of them exhibits specific properties. A paramanus does not exist in free state, rather it combines with other paramanus to form a bigger particle called anu, now known as molecule. According to him, matter consisted of indestructible minute particles called paramanus, now called atoms. (a) The idea of the smallest unit of matter was given by Maharshi Kanada. What is the contribution of the following in Atomic structure? Furthermore, as there are two unpaired electrons in an Oxygen molecule hence it is paramagnetic.Chapter 4 Atomic Structure and Chemical Bonding Class 9 - Concise Chemistry Selina double bonds formed between two oxygen atoms (O=O). As electrons are also present in antibonding molecular orbitals so weak bonds will be formed.īond order = Number of electrons in BMO – Number ofĪs the bond order in Oxygen is 2 so two bonds i.e. Out of eight electrons, six go to bonding molecular orbitals and two to the antibonding molecular orbitals. The electron filling in these molecular orbitals follows Aufbau, Pauli exclusion principle and Hund’s rule. π2py and π2pz, while two antibonding molecular orbitals i.e. 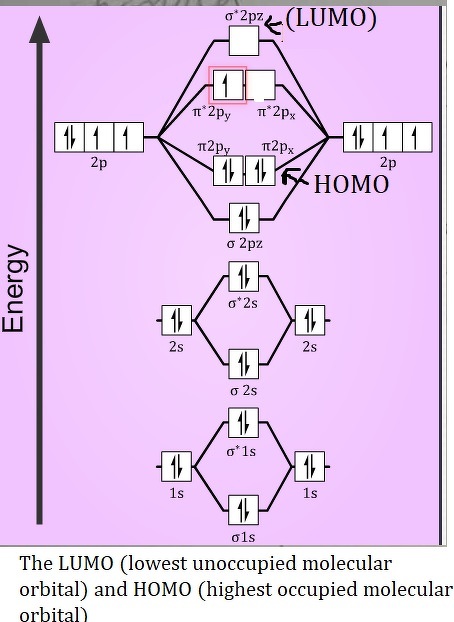
The other four p-atomic orbitals (two from each oxygen) atom combines to give four molecular orbitals, two bonding molecular orbitals i.e. Two p-atomic orbitals (one from each oxygen) atom combine to form two molecular orbitals, the bonding molecular orbital σ2px and antibonding molecular orbital σ*2px. electronic configuration of oxygen mo diagram of oxygen  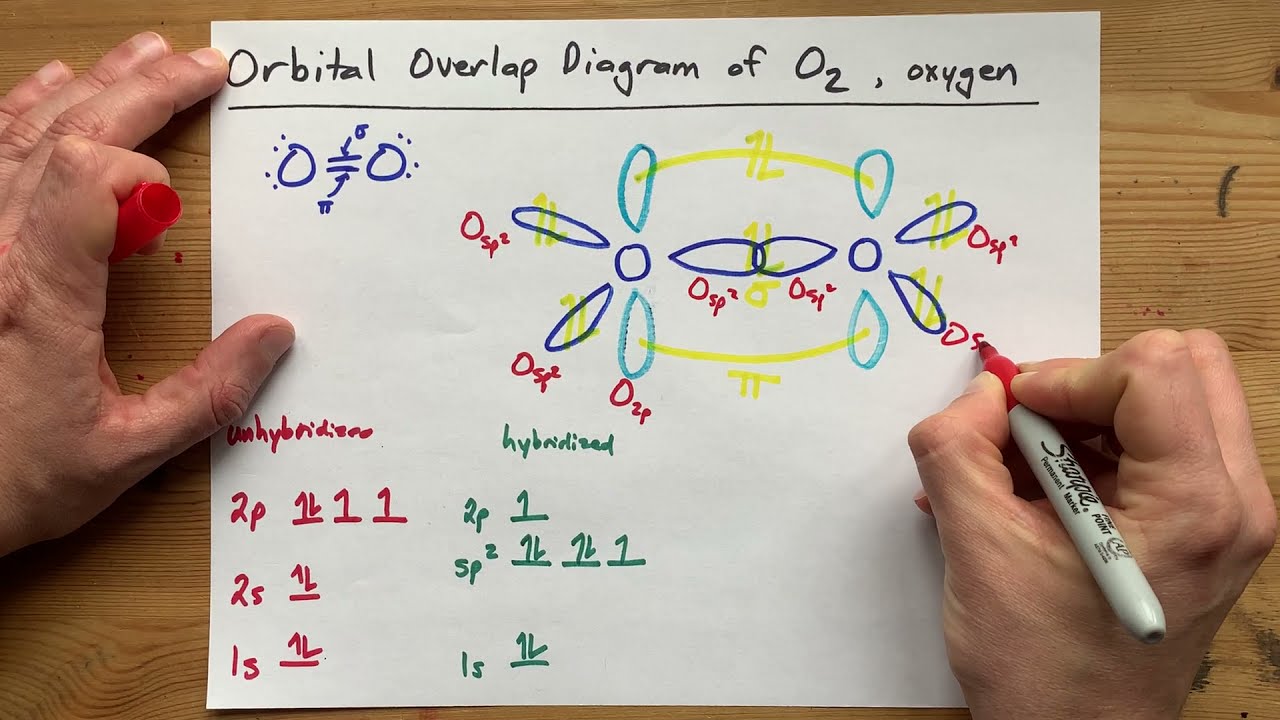
Oxygen atom has electronic configuration 1s2, 2s2, 2p4. Now let’s understand !! MO Diagram of O2 molecule mo diagram of oxygen electronic configuration of oxygen The total energy of the MOs is always equal to the total energy of combining atomic orbitals. Half of the molecular orbitals (MOs) having energy lower than the atomic orbitals are called bonding molecular orbitals (BMOs) while half of the molecular orbitals (MOs) having energy higher than the atomic orbitals are called Anti-bonding molecular orbitals (ABMOs) 3. Molecular orbital (MO) theory explains the construction of a molecular orbital diagram on the basis of the following main points.Ītomic orbitals(AOs) linearly combine with each other to form an equal number of molecular orbitals (MOs). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
